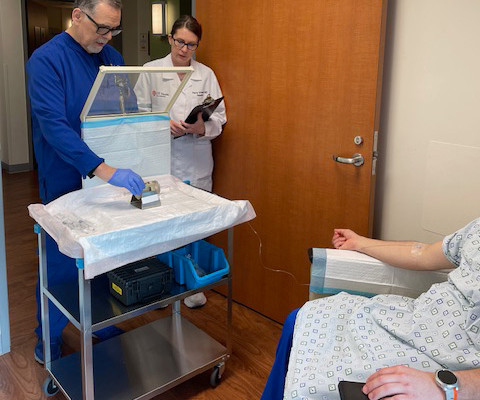
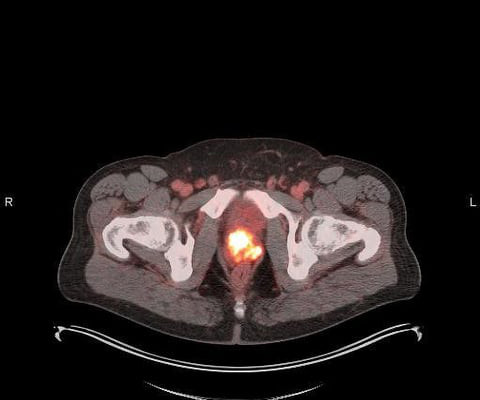
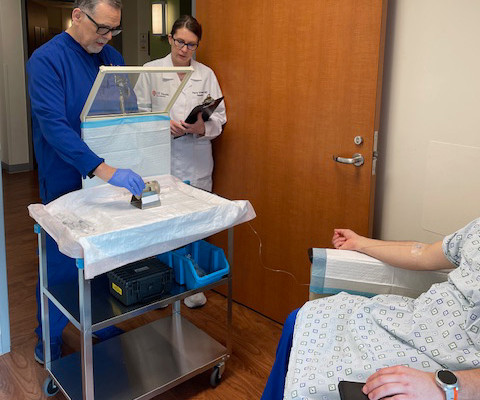
First Participant Treated Using NorthStar Medical Radioisotopes’ Electron Accelerator-produced Copper-67 (Cu-67) in Clarity Pharmaceuticals’ Phase I/IIa Theranostic Clinical Trial Investigating Cu-67 SARTATE for Treatment of Neuroblastoma
Imaging Technology
AUGUST 29, 2023
Administration of the therapeutic dose took place as part of an ongoing Phase I/IIa theranostic clinical trial conducted by Clarity Pharmaceuticals to investigate the safety and efficacy of Cu-67 SARTATE in pediatric patients with high-risk neuroblastoma.











Let's personalize your content